
In 1913, the Bohr's model was a giant leap forward because it incorporated features of the newborn quantum mechanics into the description of atoms and molecules. Nuclei were positive electric, with various masses but much larger than electrons, yet very small in size." "Ernest Rutherford discovered the nucleus in 1911. "Electrons were found to be negative electric, and all with the same mass and very small compared with atoms," says Dudley Herschbach, a Harvard chemist who shared the Nobel Prize in Chemistry in 1986 for his "contributions concerning the dynamics of chemical elementary processes," in an email. 
His best guess was the " plum pudding model," which depicted the atom as a positively-charged pie studded with negatively-charged areas scattered throughout like fruit in an old-timey dessert. Thomson just hypothesized that electrons existed, but he couldn't work out exactly how electrons fit into an atom. Thomson discovered electrons - negatively-charged particles inside the atoms everyone had spent the better part of a century believing were entirely indivisible - as the smallest things that existed. Scientists suspected atoms existed for a long time before they could conceptualize their structure - even the ancient Greeks figured the matter of the universe was made up of components so small they couldn't be broken down into anything smaller, and they called these fundamental units atomos, which means "undivided." By the end of the 19th century, it was understood that chemical substances could be broken down into atoms, which were very small and atoms of different elements had a predictable weight.īut then, in 1897, British physicist J.J. But we've got an estimation of what a single atom looks like because of the work of a bunch of different scientists like Danish physicist Niels Bohr.Ītoms are the building blocks of matter - a single atom of any individual element is the most basic entity in nature that still abides by the rules of physics we can observe in everyday life (the subatomic particles that make up atoms have their own special rules). You can search for a picture of an atom on the internet and you'll find one, even though nobody's actually seen an atom before. Shortly after Bohr published his planetary model of the atom, several new discoveries were made, which resulted in, yet again, a revised view of the atom.Bohr's model of the atom. However, his model worked well as an explanation for the emissions of the hydrogen atom, but was seriously limited when applied to other atoms. Moving up the ladder increases your potential energy, while moving down the ladder decreases your energy.īohr's work had a strong influence on our modern understanding of the inner workings of the atom. As you move up or down a ladder, you can only occupy specific rungs and cannot be in the spaces in between rungs. An everyday analogy to the Bohr model is the rungs of a ladder. The electron is not allowed to occupy any of the spaces in between the orbits. The orbits that are further from the nucleus are all of successively greater energy. The ground state of the hydrogen atom, where its energy is lowest, is when the electron is in the orbit that is closest to the nucleus. When the electron is in one of these orbits, its energy is fixed. It accounts for a wide range of physical phenomena, including the existence of discrete packets of energy and matter, the uncertainty principle, and the exclusion principle.Īccording to the Bohr model, often referred to as a planetary model, the electrons encircle the nucleus of the atom in specific allowable paths called orbits. This is a theory based on the principle that matter and energy have the properties of both particles and waves. This was the basis for what later became known as quantum theory. When the energy is removed, the electrons return back to their ground state, emitting a corresponding amount of energy-a quantum of light, or photon. 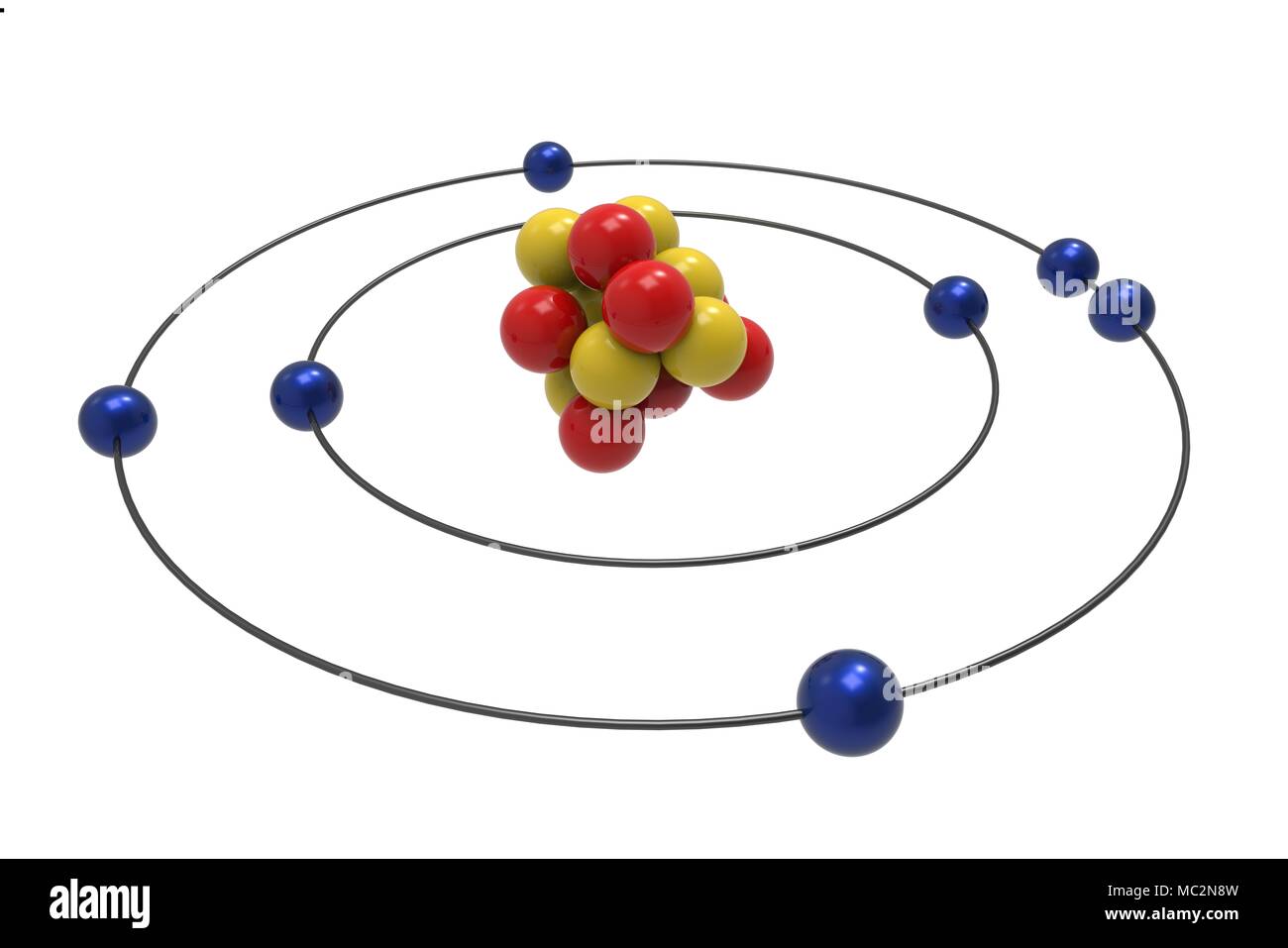
(Credit: Zachary Wilson Source: CK-12 Foundation License: CC BY-NC 3.0(opens in new window))īohr explained that electrons can be moved into different orbits with the addition of energy. \): Bohr's atomic model hydrogen emission spectra.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
